Site Evaluation Summary
Site Evaluator: Shu Qiu, PA-C
With Shu being both my preceptor and site evaluator for this rotation, she did an incredible job of helping me become more comfortable with the surgery service. While we worked together on the general surgery service, we met twice to discuss specific cases. The first case involved a patient with a recurrent small bowel obstruction where we also review 10 pharm cards. Our second meeting focused on a patient I followed on the neurosurgery service with a traumatic brain injury. Shu has worked in many different positions in her medical career but has been on the surgery service the longest. She used her knowledge to help my H&P writing improve specifically in formulating a plan for my patients. Being that this was my last rotation and I will be writing for my own patients in the near future, she really focused on the concept that when you write your H&P, a provider that will come across your patient should be able to read your H&P and completely understand the patient’s hospital course. Ensuring that you have a sound plan also makes sure that there are no gaps or misinterpretations to how you want your patient to be treated.
Rotation Reflection
I was exposed to a wide range of different doctors and residents during my surgical rotation. I rotated on the general surgery service for three weeks, urology for one week, and neurosurgery for one week. This variety made it possible for me to learn from a wide range of instructors and to have a well-rounded experience.
One of the best aspects of the surgery rotation is being exposed to performing multiple procedures that I had not yet had in my clinical year. Suturing, knot tying, placing NG tubes, and wound care were among the many skills I was performing on a near-daily basis.
This rotation pushed me and improved my anatomical knowledge. I feel much more at ease than I ever have with my knowledge of muscle groups, vasculature, and landmarks after five long weeks. I recall that when I watched my first laparoscopic cholecystectomy, I had no idea what I was seeing or what was happening. However, at the end of the rotation, I even found myself going through the steps of the case in my head before they happened. It was also a plus that our board review class came immediately before this rotation because it helped me prepare for a lot of what I saw in the operating room and will see on my end-of-rotation exam.
Each day when I received the schedule of surgeries I would be scrubbing into, I referred to my copy of “Surgical Recall” to refresh myself on the most important anatomical structures and clinical pearls of the disease process/surgery being performed. This helped me feel more confident when an attending surgeon or resident would ask me questions related to the case.
While the hours of this rotation were the most challenging of the clinical year, I used this time to review lab work, understand when particular labs need to be ordered, and home in on my skills of presenting patients during rounds.
Overall, surgery was an excellent rotation for me because it helped clarify my professional goals and helped me prepare for my boards in just a few short weeks.
Journal Article and Summary
Article: Effectiveness of Antiseizure Medications Therapy in Preventing Seizures in Brain Injury Patients: A Network Meta-Analysis
Citation: Huo, Xianhao, et al. “Effectiveness of Antiseizure Medications Therapy In Preventing Seizures In Brain Injury Patients: A Network Meta-Analysis.” Frontiers in Pharmacology, vol. 13, 15 Sept. 2022, https://doi.org/10.3389/fphar.2022.1001363.
- Traumatic brain injury (TBI) is mainly caused by direct or indirect external forces on the head. More than 50 million people worldwide suffer from TBI each year. These injuries can result in various disabilities, including neurological deficits, memory loss, and other negative results, making TBI a chronic health condition and a global healthcare burden.
- Post-traumatic epilepsy (PTE) is a recognized complication of TBI. Depending on the location and severity of the bleeding, PTE can occur immediately within 24 h after trauma, early within the first 7 days following trauma, and late after 7 days following trauma.
- Over the past 30 years, the cumulative incidence of PTE was 2% for mild brain injury, 4% for moderate brain injury, and 15% for severe brain injury. Prophylactic treatment with anti-seizure medications is increasingly being used to reduce the risk of developing PTE following TBI.
- In order to assess the efficiency of anti-seizure drugs in avoiding early or late seizures in TBI patients, a ranked network meta-analysis was conducted in this study. They also assessed the mortality rate and unfavorable side effects associated with the various regimens.
History and Physical – Rotation 9
Identifying Data
January 26th, 2022 – 6:00AM
CP, 37M, Hispanic, Queens, NY
Informant: Chart, ED staff
Referral Source: EMS
Chief Complaint: Found unconscious on sidewalk
History of Present Illness
CP is a 37-year-old male with PMH of ETOH abuse and history of multiple recent falls, latest 12 days ago and was admitted to Elmhurst Hospital with traumatic subdural hematoma, BIBEMS presented to the ED today after being found on the sidewalk with altered mental status. When patient was brought to ER, he was intoxicated, mumbling but moving all extremities with good strength. Patient was later brought to CT Head and after CT scan, the patient started to deteriorate, and patient was intubated in CEU. Neurosurgery was consulted after patient was intubated. CT head showed a large intraparenchymal hemorrhage and adjacent hemorrhages centered in the left temporal and posterior left frontal lobes, spanning a region of up to 9 cm, with surrounding edema. Mild subarachnoid hemorrhages in these regions as well with small subdural hematoma at the left temporal convexity overlying the left intracranial hematoma.
Past Medical History:
- Right subdural hematoma 1/2023 – received TXA and Keppra
- Interhemispheric fissure subarachnoid hemorrhage 6/2022 – received TXA and Keppra
Past Surgical History: Unknown
Medications: Started in hospital. Home medications unknown.
- Acetaminophen 975mg, Orogastric tube, Q6h
- Enoxaparin 40mg, SC, nightly
- Famotidine 20mg, orogastric tube, Q12h
- Levetiracetam (Keppra), 1,000mg, IV Q12h
- Propranolol 20mg, orogastric tube Q8h
- Thiamine 100mg, orogastric tube daily
- Lorazepam PRN
Allergies: Unknown
Family History: Unknown
Social History: +EtOH abuse, remainder unknown
Review of Systems: Unable to obtain due to intubation at the time of consult
Physical Exam:
General: 37M, patient intubated and sedated in CEU, unable to follow any commands, awaiting transport to OR
Vitals:
- BP(Seated): R – 131/78 [113/133]/[52-78]
- P: 70 [65-104]
- R: 14, ventilated
- T: 38.1C [37.8-38.5]
- O2 Sat: 100% ventilated [96-100]
- Height: 65 inches – Weight: 137.8lbs – BMI: 22.78 – Normal
General RASS score: 0
HEENT: Orally intubated, OGT in place, left pupil 4mm, sluggish, right pupil 2mm, sluggish
Cardiovascular: regular rate & rhythm
Respiratory: Ventilating via ETT
Abdomen: Soft, nondistended
GU: Foley in place with light yellow urine
Musculoskeletal: Moves left sided extremities purposefully to noxious stimuli. Minimal withdraw to RLE to noxious stimuli. No movement to RUE
Neuro: GCS E(1)V(NT)M(4)
Associated Imaging:
CT Head was done which showed a large IPH and adjacent hemorrhages centered in the left temporal and posterior left frontal lobes, overall spanning a region of up to 9 cm, with surrounding edema. Intramural hematoma anteriorly inferiorly in the right frontal lobe, overall spanning a region of approximately 2.8 cm, surrounding hematoma. Mild subarachnoid hemorrhages in these regions as well with small subdural hematoma at the left temporal convexity overlying the left intracranial hematoma.
Associated mass effect, including lateral shift of midline structures, medialization of the uncus, and findings suspicious for early/incipient herniation. Neurosurgery consulted for the above findings.
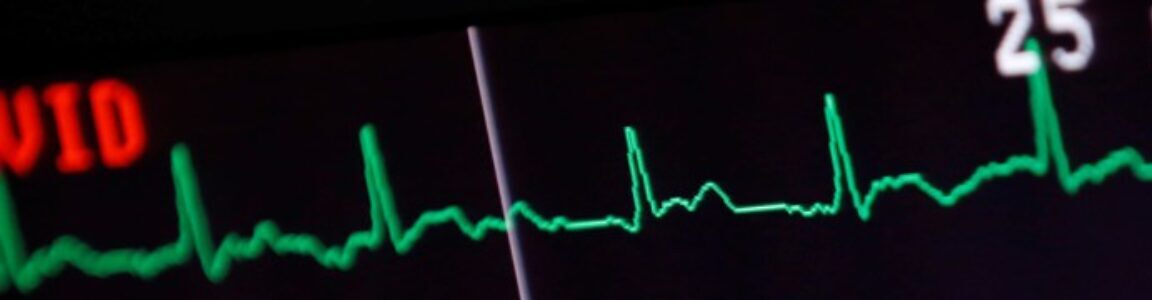
Labs:
BMP:
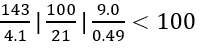
CBC:
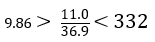
T&S: O+
ABG
- pH 7.32
- PCO2 48
- HCO3 24.4
Prothrombin Time: 13.0 (10.0 – 13.0 seconds)
INR: 1.12 (0.87 – 1.13)
Ethanol Level: 294.2
- 50-100 mg/dL: Flushing, slowing of reflexes, impaired visual acuity
- > 100 mg/dL: Depression of CNS
- > 400 mg/dL: Fatalities reported
Assessment:
37M with history of EtOH abuse and several falls with TBI presented on 1/25/23 with intoxication and large left temporal IPH yielding midline shift and uncal herniation along with right IPH, L subdural hematoma, subarachnoid hemorrhage, required intubation and was taken to OR for left craniectomy and R EVD placement.
Plan
#TBI
- Book as Level 1 for left hemicraniectomy for decompression, possible ICH evacuation. Right frontal EVD placement.
- Repeat CT head 1/27
- HOB > 30 degrees
- Monitor ICP and CPP via EVD
- Monitor EVD outputs
- Continue Keppra 1g BID
#EtOH Abuse
- Continue Thiamine 100mg daily
- Monitor electrolyte abnormalities
#Hypertension
- Continue Propranolol
- Continue hemodynamic monitoring
#Acute blood loss anemia
- Trend hemoglobin, transfuse PRN
#DVT prophylaxis
- Lovenox 40mg injection SC nightly
PANCE Prep Plan
Typhon Logging
Curriculum Vitae & Cover Letter
Site Evaluation Summary
Site Evaluator: Carlos Melendez, PA-C
As Professor Melendez is employed at Woodhull Medical Center, he was both the preceptor and site evaluator for this rotation. Being that he was on-site, we were able to have our site evaluation in person. My patient presentation involved a woman presenting to L&D triage for feeling decreased fetal movement. His comments on my focused history and physical were taken constructively and we spoke about the occurrence and management of decreased fetal movement. Because this meeting was later in my rotation, I also presented a systematic review and meta-analysis that looked at perinatal mortality when mothers were instructed to practice fetal movement counting. This practice often called kick counting, is a way a mother can help monitor the movements of her unborn baby by counting the number of kicks in a certain time period. While there are weak associations with some secondary outcomes such as preterm delivery, induction of labor, and cesarean delivery, there were no statistically significant improvements in pregnancy outcomes. As we were discussing labor and delivery, we took some extra time to understand the management of expectant mothers and the different roles that midwives, nurses, PAs, and physicians play in labor and delivery.
Rotation Reflection
Of all of the rotations this year, this was the most apprehensive I was prior to starting. As a male student, it is not uncommon for patients to be uncomfortable in this setting, especially when performing exams for gynecological complaints. Fortunately, having a male preceptor for this rotation, I was able to get more comfortable with ensuring patients that their care, safety, and respect are the highest priority.
In terms of experiences, I appreciated that as Woodhull Medical Center is a part of the NYC Health&Hospitals network, it provided me with the opportunity to interview a wide variety of patient populations and allowed me to practice a wide range of clinical skills in a safe teaching environment.
One area that I had not considered as a challenge going into this rotation was your ability to connect with a patient on a personal level. As one of my preceptors told me, in OBGYN, you are going to see mother’s on the happiest and saddest days of their lives. It is our job as providers to show sympathy and compassion while also treating our patients however they may need.
My nights on the labor and delivery unit were a great chance for me to put myself out there and be involved, whether that meant introducing myself to each of the patients and their families, so they are comfortable with my presence as a student or making sure the different providers knew I was interested in being involved in each case, looking for opportunities to develop new skills.
The two weeks I spent in the clinic during this rotation were perhaps my favorite. It was quite instructive to be able to spend an entire day with a PA or midwife and observe how they handle each patient. I was able to gain the most practice in procedures including using the speculum, performing pelvic and breast exams, and collecting culture samples. Each provider always seemed eager to share their knowledge and took great care to ensure that each patient was at ease while gradually letting me take on more responsibility throughout the visit.
Overall, I had a great time on this rotation, as each week there were different opportunities to be involved directly with patient care and to work as a part of a cohesive team that is essential in providing the highest standard of care. It also reinforced the notion that healthcare is a team approach; and seeing so many providers with different levels of training work seamlessly together, you could see that patient care is the top priority.